|
Although the outcome is an approximation, it's provided with sufficient precision. We use scientific notation to express all of the small values more clearly. Where the temperature T is in ☌, and the values of coefficients are the following: To get around this, our water density calculator uses an approximate equation based on the 5 th order polynomial: While there are tables of pure water density between water's freezing point ( 0 ☌ or 32 ☏) and its boiling point ( 100 ☌ or 212 ☏), there isn't a straightforward formula which yields the exact value for a given temperature. Water has an intriguing property – it reaches its maximum density at about 4 ☌ or 40 ☏. Qualitatively, it's fairly simple, but from a mathematical point of view, estimating the outcome is a different story. 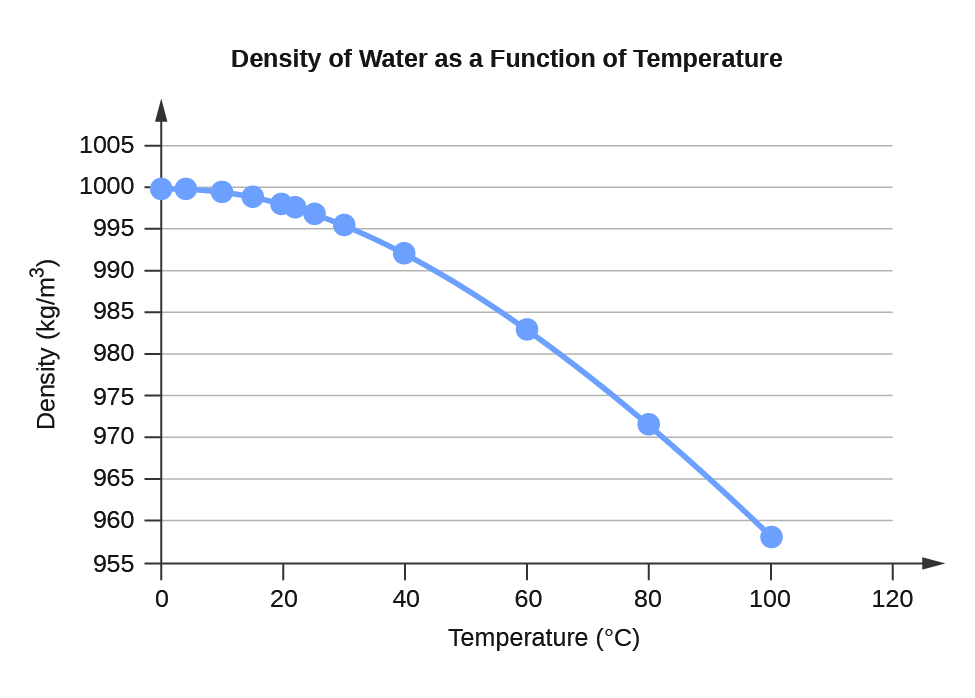
As a result, the mass stays constant, but the volume increases, causing the density to decrease. 
Of course, this tale is disputed today because Archimedes was not precise in all his measurements, which would make it hard to determine accurately the differences between the two crowns.You probably know of the phenomenon of the density of water changing at different temperatures, even if you've never thought about it in scientific terms ever wondered why ice floats? This change is mainly caused by thermal expansion – where the same amount of substance occupies more and more space as the temperature increase. The king's "gold" crown, therefore, was not made of pure gold. The king's crown displaced more water than the gold crown of the same mass, meaning that the king's crown had a greater volume and thus had a smaller density than the real gold crown. He then realized that he could use a similar process to determine the density of the crown! He then supposedly ran through the streets naked shouting "Eureka," which means "I found it!" in Latin.Īrchimedes then tested the king's crown by taking a genuine gold crown of equal mass and comparing the densities of the two. While meditating on this puzzle in a bath, Archimedes recognized that when he entered the bath, the water rose. Archimedes did not know how to find the volume of an irregularly shaped object such as the crown, even though he knew he could distinguish between elements by their density. The story goes that Archimedes was asked to find out for the King if his goldsmith was cheating him by replacing his gold for the crown with silver, a cheaper metal. The Greek scientist Archimedes made a significant discovery in 212 B.C. For example, it explains how hot air balloons work. This explains why ice floats to the top of a cup of water: the ice is less dense.Įven though the rule of density and temperature has its exceptions, it is still useful. With this volume increase comes a decrease in density. As the water freezes into the hexagonal crystals of ice, these hydrogen bonds are forced farther apart and the volume increases. 
In the water molecule, the hydrogen bonds are strong and compact. Ice is less dense than water due to hydrogen bonding. This is unusual as solids are generally denser than their liquid counterparts. Looking at the table, you can also see that ice is less dense than water. Liquid water also shows an exception to this rule from 0 degrees Celsius to 4 degrees Celsius, where it increases in density instead of decreasing as expected. \), the density of water decreases with increasing temperature.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
